|
The unprocessed form of silicon is used in the making of silica sand, clays, and stone.įor Portland, which is the most common type of cement, silicates are used. Silicone rubber is used in waterproofing systems in bathrooms, roofs, and pipes.īeing a semiconductor, silicon is used in transistors and solid-state devices including microelectronics and the computer industry. Silicon is used to make polymers of silicon-oxygen with the methyl group attached known as silicones and its oil is a lubricant added in hair conditioner and cosmetics. Silicon is used to make many alloys such as aluminium–silicon and ferrosilicon known as iron–silicon which is widely used in the steel industry. Silicon is used in the production of ceramics, bricks, and fire bricks. Most silica gels are found to occur in an aqueous state. Silicic Acids:- Hydrated silica gels are formed as a result of increasing water concentration. Unlike carbon tetrahalides, these readily hydrolyze in water. Halides:- Silicon compounds, mostly carbides, form silicon tetrahalides when they react with stable halogens. Silica:- Silica (Silicon dioxide) mainly consists of granite and sandstone which is used in the manufacture of glass, abrasives, in water filtration, as a food additive, and many more. Silicon carbide is also known as Carborundum which is widely used as powdery material to polish or grind other material and has refractory properties. Due to their similar structure, the heat of formation of these elements is similar. Silicides:- Silicides have similar structures to borides and carbides. When exposed to air, these polymers are very reactive and can easily catch fire. Silanes:- Silanes are very strong reducing agents and consist of homologous series of compounds such as silicon hydrides. It also forms alloys in the molten state.Ĭompounds/Polymers of Silicon (Silicon Materials) Silicon combines with oxygen, phosphorus, nitrogen, and other elements. Silicon is very reactive at higher temperatures. Being solid, it does not combine with oxygen or other most related elements. Silicon is very similar to metals in terms of chemical behaviour.Īt room temperature, silicon is a relatively inactive element. 
Silicon is purely electropositive in its chemical behaviour, has a metallic lustre, and is considered to be very brittle. Silicon in its purest form is an intrinsic semiconductor Although adding impurities in small amounts helps in highly increasing the intensity of semiconductor. The silicon melting point is 1,410☌ and the silicon boiling point is 3,265☌. The density of silicon is 2.3296 grams per cubic centimetre. The atomic number of silicon is 14 and its relative atomic mass is 28.085 u. One silicon's allotrope is in the form of needle-like, shiny, greyish-black crystals or flat plates, while the other one has no crystal structure and it exists usually as a brown powder. Allotropes are the element forms which have various chemical and physical properties. The minerals which contain silicon dioxide are known as silicates.īeing a metalloid, silicon also occurs in two allotropic forms. Silicon exists in nature in many dioxide forms, making compounds with oxygen, phosphorus, magnesium, and others. About 97% of Earth's crust is made of rocks and comprises silicon and oxygen compounds. It occurs in combined form in nature as silicon dioxide which is commonly known as silica. Silicon is the second most abundant electropositive element which makes up 27.7% of the Earth's crust by mass. Silicon is, for the most part, unreactive. Silicon is a hard yet breakable crystalline solid, and it has a bluish-grey metallic lustre, and also Silicon is tetravalent, that is to say, its valency is 4, it is also a semiconductor. Electronic configuration of silicon is (Ne) 3s 2 3p 2. Silicon used in electronics acts as metals, while the glass which is a silicon compound has non-metallic qualities. 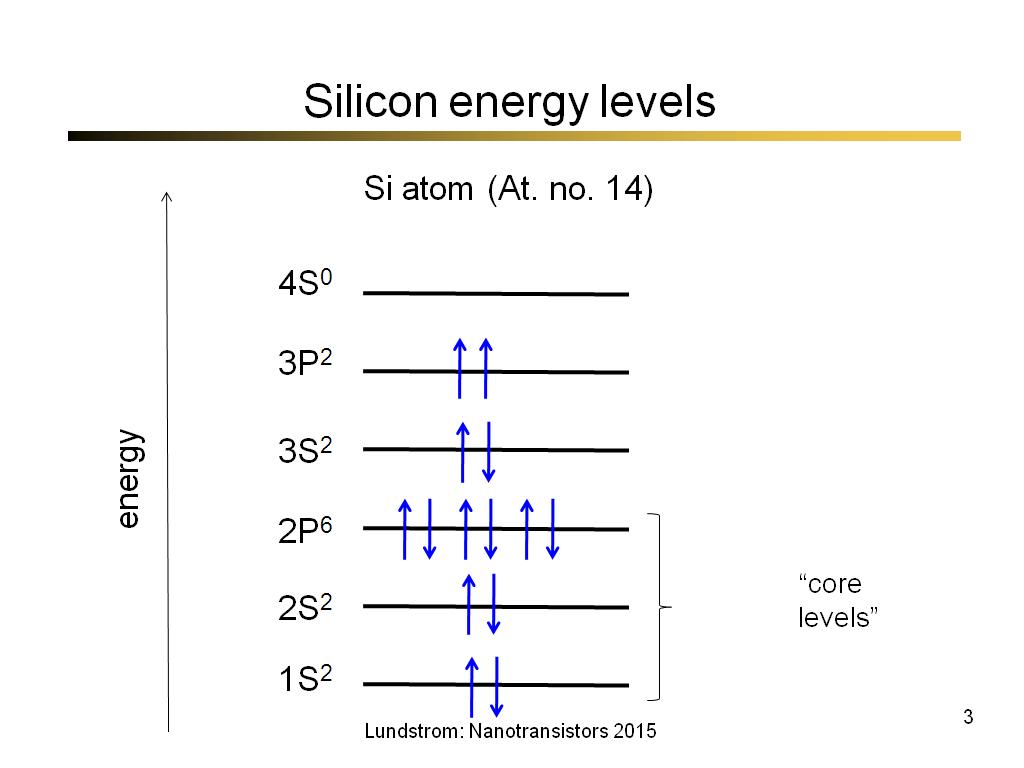
Silicon is metallic, one of the seven elements which have both the characteristics of non-metal and metal depending upon the other element to which it combines. Carbon is above it, while the elements like germanium, tin, lead, and flerovium are below it. Silicon is a member of the carbon family and is a non-metallic chemical element, having atomic number 14, and belongs to group 14, period 3 in the p-block of the periodic table. The melting point is surpassed by only boron out of all the metalloids and nonmetals. Silicates, which contain silicon and oxygen, are formed by the oxides form of the silicon. And it was because of the fact that silicon has a high chemical affinity to oxygen. Silicon was not characterized in its pure form until the year 1824 when Jons Jakob Berzelius first managed to do so.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
